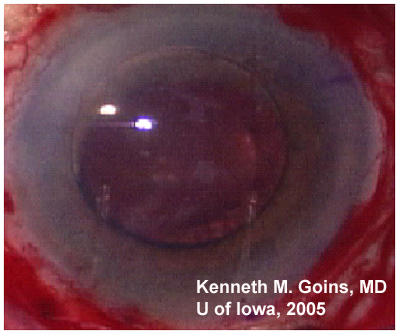
Chief Complaint: 79-year-old female referred from primary ophthalmologist for severe bullous keratopathy in the left eye.
History of Present Illness: Following cataract surgery with anterior chamber intraocular lens (ACIOL) placement in both eyes (OU) nearly 20 years before, the patient slowly developed pseudophakic bullous keratopathy first in the right eye (OD). She underwent penetrating keratoplasty (PKP) with intraocular lens exchange in the right eye in 1996. In the years following, the right eye subsequently developed corneal graft failure. The patient had an episode of anterior ischemic optic neuropathy (AION) in this same right eye that further decreased her vision.
Functioning thereafter with her left eye, the patient maintained a visual acuity that enabled her to read the newspaper and perform her activities of daily living for several years. However, the patient eventually experienced progressive decline in her vision, OS. The patient began to notice increasing tearing and light sensitivity. Vision deteriorated rapidly in the months prior to her referral to the cornea service at the University of Iowa Hospitals and Clinics.
Past Ocular History: Cataract surgery with ACIOL, OU approximately 20 years prior. Pseuophakic bullous keratopathy treated with PKP, OD and IOL exchange in 1996. Graft failure, OD. History of non-arteritic AION in the right eye (OD). Primary open angle glaucoma (POAG) being treated with Timolol drops. Closed-loop ACIOL remains in the left eye with worsening pseudophakic bullous keratopathy.
Medical History: Non-Insulin Dependent Diabetes Mellitus (diet-controlled) and hypertension.
Medications: NKDA; Timolol 2x/day (BID), both eyes (OU); Muro 128 drops (5% Sodium Chloride) 4x/day (QID) left eye (OS); Prednisolone 1%, QID, OS; and oral Losartan.
Family and Social History: Noncontributory
Ocular Exam
 |
Course: Prognosis for the right eye was very poor given the history of AION and the patient was essentially monocular. Options for restoring vision to the left eye were discussed at length with the patient including corneal transplantation with either traditional penetrating keratoplasty (PKP) or deep lamellar endothelial keratoplasty (DLEK). Prevention of further endothelial cell loss and eventual graft failure would require removal of the closed-loop ACIOL and exchange for a posterior chamber IOL.
The patient decided to proceed with DLEK and IOL exchange. The surgery was accomplished using techniques described previously by Dr. Goins at the University of Iowa (and discussed in another EyeRounds case), but with the addition of a pars plana infusion port. This infusion port had several benefits. Continuous infusion through the posterior line helped to sustain intraocular pressure and a formed anterior chamber. A formed anterior chamber allowed for easier dissection of the host endothelial button (see Video 1). Maintenance of the intraocular pressure prevented intraoperative hypotony thereby decreasing the risk of suprachoroidal hemorrhage. In addition, the pars plana infusion prevented posterior movement of air during the fixation of the donor lenticule (posterior movement of air can increase the risk of intraoperative closed angle glaucoma).
Regarding the need to transplant the patient's endothelium, DLEK seemed to be a perfect option for this patient. She had essentially normal stroma and superficial cornea; only the endothelium had been permanently damaged by her condition. DLEK allows for effective transplantation of the damaged tissue, while preserving the healthy portions of the the patient's native cornea and avoiding the additional risk and post-operative complications that can be associated with traditional PKP.
video 1. A formed anterior chamber allowed for easier dissection of the host endothelial button.
If video fails to load, use this link
Post-operative Course: One month after surgery, the patient was healing well and vision was improving. Endothelial cell counts performed by confocal microscopy confirmed nearly 2100 cells/mm2 at this point and a clear graft/host corneal interface. Five months after the surgery, the patient continued to recover appropriately, with resolving corneal edema and improvement in both visual acuity and function. Her visual acuity in the left eye (OS) had improved substantially and with minimal astigmatism. With a refraction of -1.50 +1.00 x125 the patient's visual acuity reached 20/60. This is a secondary benefit of DLEK as compared to traditional PKP. By minimizing compromise of the anterior corneal surface (no full-thickness corneal incisions and sutures), post-operative astigmatism is often minimal in DLEK patients.
Intraocular pressure was 15 mmHg in the post-operative left eye. Slit lamp examination showed a corneal lenticule in good position with a clear cornea with a thickness of 575 um by pachymetry. The anterior chamber was deep and quiet. Some peripheral anterior synechiae were noted superiorly in the angle. The posterior chamber intraocular lens (PCIOL) was noted to be in good position. Repeated confocal microscopy confirmed nearly 1700 cells/mm2 (Video 2).
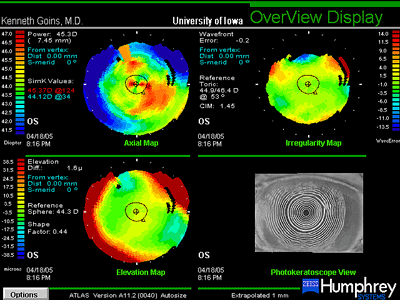
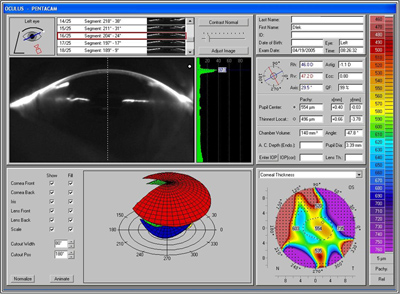
Though the 20/60 visual acuity obtained was certainly a dramatic improvement and the patient was very pleased, the cause for the maximum obtained visual acuity being thus limited was investigated further. Slit lamp examination and refraction would seem to indicate a potential beyond 20/60 (see Figure 2). Repeat measurement and refraction yielded no improvement. Optical coherence tomography (OCT) confirmed that there was no cystoid macular edema (CME). Topographic and wavefront analysis were performed. Simulated keratometry was similar to manual keratometry readings, indicating a difference of less than +1.50 diapters between the two axes. Placido image showed minimal irregularity and there was no evidence of significant higher order aberrations (HOA). Interface opacity or densiometry measurement was 37.3, above the normal of 25-30, which can be seen with lamellar transplant surgery and may somewhat hinder the best corrected visual acuity (see Figure 3). Nonetheless, the patient was very pleased with the outcome and was again able to function as she had before her visual deterioration.
Video 2: Slit lamp view, OS, after DLEK and IOL exchange
If video fails to load, use this link.
Video 3: Endothelial Cells from Video of Confocal Microscopy
If video fails to load, use this link
| A: Topographic and Placido images, OS, 4 months after DLEK with IOL exchange |
 |
| B: Pentacam results, OS, at 4 months post-operative. There is some increase in interface opacity as measured by densiometry (37.3) |
 |
The corneal endothelium is a honeycomb population of specialized cells lining the interior surface of the cornea. The cells function as pumps, maintaining corneal clarity. Death of the corneal endothelial cells results in rearrangement of the remaining cells that blanket the internal corneal surface and cellular polymorphism. The average healthy adult cornea has 2000 to 3000 cells per square millimeter (ave. 2400 cells/mm2). With aging, there is a gradual, though variable, decrease in endothelial cell count. In most patients, this natural attrition of endothelial cells never becomes clinically evident and the cornea remains clear. However, with certain types of disease (i.e. Fuch's dystrophy) or mechanical insult, endothelial cell loss may progress to a pathologically low level and corneal edema may result.
Any type of intraocular surgery, especially cataract surgery, may damage endothelial cells and hasten the decline in endothelial cell count. Pseudophakic bullous keratopathy (PBK) is a post-operative condition that can occur as a complication of cataract extraction surgery and intraocular lens placement. Both intraoperative insult to the endothelium and long-term cell damage as a result of the lens implant can lead to PBK. Proposed mechanisms for endothelial cell loss include direct trauma during surgery, prolonged irrigation, toxic medications and other agents, inflammation, or increased intraocular pressure. Anterior chamber IOLs in particular contribute to endothelial damage from "intermittent touch" (between IOL and cornea) and chronic irritation with low-grade inflammation caused by the IOL haptics or footplates. Additionally, it is hypothesized that IOLs disrupt the normal flow of aqueous in the anterior chamber which affects the nutrient flow to endothelial cells.
The loss of endothelial cells can lead to progressive stromal edema with a general decline in visual acuity. Some patients experience photophobia, haze in their vision that is worse in the mornings, and tearing. As edema increases, subepithelial bullae may form and rupture, causing the patient significant bouts of painful micro-defects in the corneal surface. Early PBK may be manifest in the immediate post-operative period or symptoms may not present for many years. Some studies have shown that endothelial cell loss may continue to progress for many years after the operation. One study followed patients for up to seven years after surgery and showed progressive endothelial cell loss greater than that seen in control patients. Particular attention has also been directed at the relationship between intraocular lens type and the severity of endothelial cell loss. Many studies report that anterior chamber intraocular lenses are associated with a greater degree of endothelial cell loss than posterior chamber intraocular lenses.
Mild PBK may be managed with hypertonic saline drops and ointments that help dehydrate the edematous cornea. Eventually, however, the endothelium may need to be replaced. Traditionally, this required a transplant of the entire cornea. More recent surgical techniques make it possible to transplant an inner lamella of cornea with healthy endothelium (see Video 1).
EPIDEMIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT
|
Suggested citation format: Wynn P, Graff JM, Goins KM: Pseudophakic Bullous Keratopathy: Deep Lamellar Endothelial Keratoplasty (DLEK) and Intraocular Lens (IOL) exchange with Anterior Vitrectomy. Eyerounds.org. November 17, 2005; Available from: http://www.EyeRounds.org/cases/40-Pseudophakic-Bullous-Keratopathy-DLEK-w-IOL-exchange.htm.

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.