Poor vision and pain in the left eye
A 51-year-old male with keratoconus and history of rigid gas permeable (RGP) contact lens wear in the left eye presented with left eye redness, pain, light sensitivity, and tearing for 1 week. He was initially seen at an acute care clinic where he was told he had scratched his conjunctiva. He then presented to his regular ophthalmologist who found him to have acute corneal edema and referred him to the University of Iowa Hospitals and Clinics (UIHC). He was started on ciprofloxacin and hypertonic saline drops by the outside provider. At the time of his presentation, his pain was rated as 7 out of 10 and described as a constant surface irritation and scratchy sensation. He described constant tearing and blurry vision which did not change throughout the day. The patient took out his contact lens when symptoms began.
 |
 |
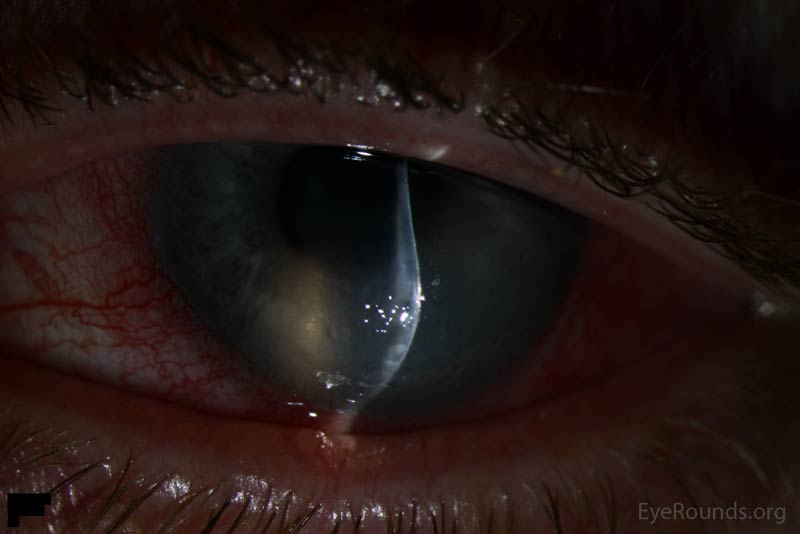 |
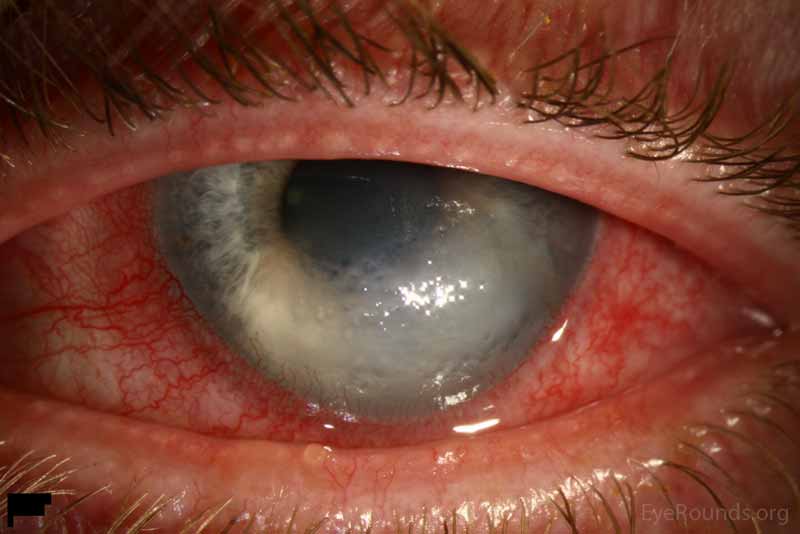
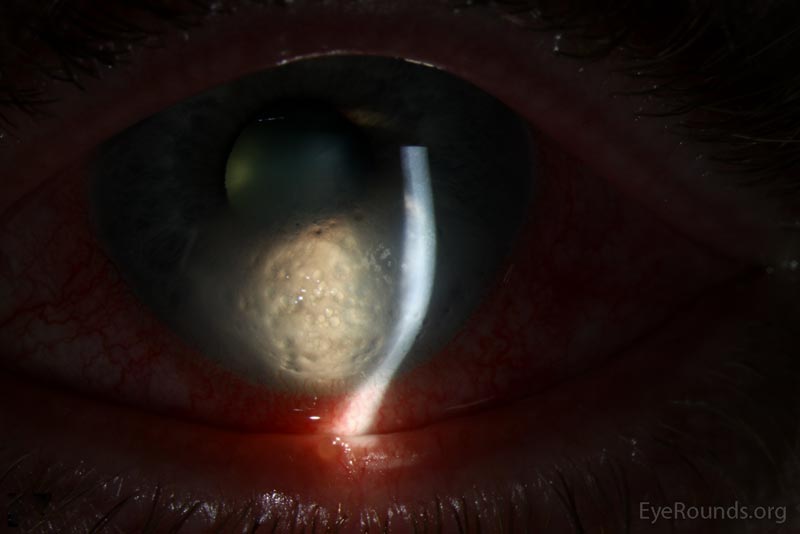
Acute corneal hydrops in the setting of keratoconus
Initial conservative management included prednisolone 4 times daily, sodium chloride 5% (Muro 128) drops 4 times daily, and cyclopentolate 2 times daily in the affected eye. A Kontur bandage contact lens was placed for comfort. The patient was placed on timolol once daily to treat the ocular hypertension caused by reactive inflammation.
Placement of an anterior chamber sulfur hexafluoride (SF6) gas bubble was offered to speed his recovery, but due to upcoming travel necessitating air travel and inability to position due to work requirements, he declined.
The edema gradually improved and 3 weeks later he was found to have some improvement in corneal clarity, but a large amount of scarring and contour irregularity in the area of the hydrops. A penetrating keratoplasty (PK) was performed for restoration of vision.
Acute corneal hydrops is caused by the acute disruption of Descemet's membrane in the setting of corneal ectasia. Hydrops is a term used to denote an abnormal accumulation of fluid in a body tissue or cavity. The term is also used to describe fetal swelling in utero, often due to Rh blood group isoimmunization (hydrops fetalis). Acute corneal hydrops occurs in approximately 3% of patients with keratoconus. Although most cases of hydrops are associated with keratoconus due to the prevalence of the disease, the incidence of hydrops is actually higher in other corneal ectatic disorders such as pellucid marginal degeneration and keratoglobus, with some reports being as high as 11% [1-2]. The average age of onset is typically around 25 years of age, with males being more commonly affected than females [3]. A history of eye rubbing and seasonal allergies is associated with hydrops development [1,3].
The focal corneal edema of acute hydrops results from the compromise of the barrier function of Descemet's membrane and subsequent fluid uptake by the overlying corneal stroma. It has been postulated that resolution of hydrops requires two steps. First, the detached Descemet's membrane must reattach to the posterior stroma. This process is dependent on the depth of the detachment. Second, the endothelium must migrate from the reattached Descemet's membrane to cover the gaps between the broken ends. This process depends on the size of the break [4].
Most cases of acute corneal hydrops spontaneously resolve over 2-4 months [5-7]. Depending on the degree of swelling and timeline of resolution, vision-impairing scarring can necessitate the need for corneal transplantation. Larger disruptions of Descemet's membrane are associated with more prolonged resolution of corneal edema, increased risk of neovascularization, and a worsened visual outcome after final resolution of edema compared to smaller disruptions of Descemet's membrane [8].
Clinical manifestations of acute corneal hydrops include severe corneal edema with a corresponding reduction in visual acuity. Epiphora, photophobia, and pain may also occur. If these manifestations are seen in patients with previously diagnosed corneal ectasia and/or evidence of corneal ectasia, the diagnosis of acute corneal hydrops can be made [9].
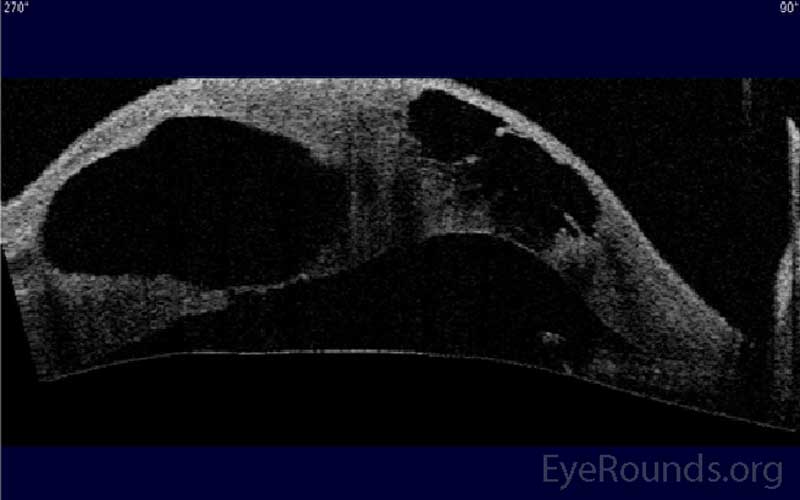
Anterior segment optical coherence tomography (AS-OCT) is not required for diagnosis but can be helpful to quantify the extent and location of corneal edema and the extent of the break in Descemet's membrane. Imaging techniques such as AS-OCT also allow improved ability to monitor the response to treatment [9].
 |
Due to the spontaneous resolution of acute corneal hydrops, classical treatment is focused on medical therapy to increase patient comfort and prevent permanent sequelae. Conservative management for acute corneal hydrops includes hypertonic sodium chloride to reduce epithelial edema, and cycloplegia for comfort. Topical steroids are controversial, but we often employ them to reduce the inflammation and subsequent neovascularization that can accompany these episodes. A bandage contact lens can be placed for comfort, but a large diameter soft lens may be necessary to fit the steep contours of these eyes.
Pneumatic descemetopexy, or placement of an anterior chamber air or gas bubble to tamponade the Descemet's membrane break, has been shown to speed recovery following acute hydrops [5-7]. In the last 15 years, this practice has gained popularity because of its ability to significantly reduce the time needed for the resolution of edema. The quicker recovery not only shortens the period of discomfort and poor vision for the patient, but theoretically decreases the risk of visually-significant scarring and need for post-resolution corneal transplantation [5-7,9]. Miyata and colleagues found that the corneal edema lasted an average of 20 days in a group of 9 patients receiving intracameral air compared to 65 days for a control group of 21 patients. The treatment group was able to return to hard contact lens use in one-fourth the time of the control group. However, there was no difference in final best corrected visual acuity after the complete resolution of edema [5]. Panda and colleagues showed that corneal hydrops resolved at an average of 4 weeks in 9 patients who received injections of intracameral SF6 gas compared to 12 weeks in the control group of 9 patients. This study showed that the treatment group had slightly better best corrected visual acuity, but it was measured at 12 weeks and not after resolution of edema [6]. Basu and colleagues, using C3F8 gas and a much larger group of patients, showed that the edema resolved significantly quicker in the treatment group (57 days for 62 eyes vs. 104 days for 90 control eyes (p<0.0001)). Notably, best corrected visual acuity was not significantly different between the two groups [7]. Ting and Srinivasan reported using C2F6 gas with a similar increase in the rate of edema resolution to the previous studies [10]. No study has yet found a difference in the need for corneal transplantation between those who received a gas or air bubble and those who did not. Several groups have reported using an air bubble along with sutures through Descemet's membrane to even more tightly re-adhere the membrane after hydrops [11-12].
The choice between air and gas depends on the estimated amount of time needed to repair the defect in Descemet's membrane. Air lasts the shortest duration, often only 2-3 days, which may require repeat bubble placement to achieve the desired effect. This was seen in the study by Miyata et al. where 78% of patients required the placement of more than one bubble [5]. At our institution, SF6 is normally utilized with the duration of 7-10 days typically being sufficient to allow for re-apposition and healing of Descemet's membrane.
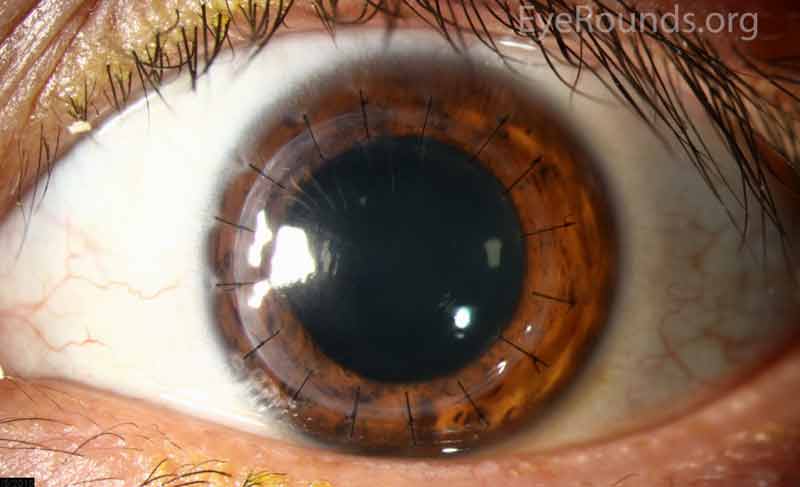
Penetrating keratoplasty (Figure 4) can be performed to treat the underlying ectatic disorder and any corneal stromal scarring that may result from the hydrops. Deep anterior lamellar keratoplasty (DALK) is extremely challenging to perform following hydrops due to difficulty separating Descemet's membrane from the posterior stroma in the setting of a Descemet's break and the posterior scarring that often accompanies these episodes. Thus, PK is usually our procedure of choice for these patients. When reviewing PK outcomes in ectatic patients who previously developed hydrops, studies have been inconclusive regarding differences in graft survival between grafts with prior episodes of hydrops and grafts without prior hydrops [13-14]. Basu et al. recently compared the allograft survival in the absence of rejection for keratoconus eyes with and without hydrops and found eyes with hydrops had significantly decreased graft survival (83% 5 year rejection-free graft survival in 32 hydrops eyes vs. 98% in 70 non-hydrops eyes) [15]. The proposed mechanism for this increase in graft rejection and failure rates is the presence of intraocular inflammation that accompanies hydrops. While corneal graft success rates decrease somewhat following hydrops, these patients still often have excellent postoperative outcomes.
 |
Treatment/Management/Guidelines
EPIDEMIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT/MANAGEMENT
|
Greenwald MF, Vislisel JM, Goins KM. Acute Corneal Hydrops. EyeRounds.org. August 3, 2016; Available from: https://eyerounds.org/cases/241-acute-corneal-hydrops.htm

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.