Eye strain, blurry vision, and headaches while reading.
A 15-year-old male presented to the orthoptic clinic at the University of Iowa Hospitals and Clinics (UIHC) for evaluation of worsening headaches and eye strain while reading, which had been present since he started high school the previous year. He complained of blurry vision while reading and letters running together on the page. He often had to cover one eye to read comfortably.
Myopia
Attention deficit hyperactivity disorder (ADHD)
Albuterol inhaler
Cetirizine
Guanfacine
Non-contributory
Freshman in high school
Negative
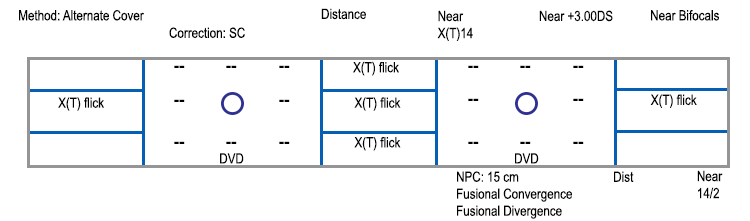
Convergence insufficiency
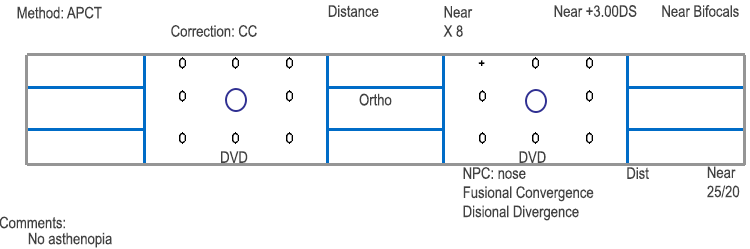
The diagnosis of convergence insufficiency (CI) was made based upon: (1) exodeviation greater at near than distance, (2) reduced convergence amplitudes, and (3) remote near point of convergence (NPC). Pencil push-up exercises were initiated in order to normalize the NPC. At his 3 week follow-up visit, the NPC was found to be within normal limits (converging to the nose) and prism recession exercises were initiated using a 12 prism diopter base out prism. He worked through prisms until he was able to converge through a 25 prism diopter base-out prism. After completing the exercises, at his 8 month follow-up visit the NPC remained normal, his fusional convergence amplitudes had increased, and there was complete resolution of asthenopia. He also gained better control of his intermittent exodeviation at near: the angle of the deviation decreased from 14 to 8 prism diopters and it went from being an intermittent exotropia to an exophoria.
Convergence insufficiency (CI) affects 2.25-8.3% of the population and is characterized by an inability to converge the eyes smoothly as the object of regard moves from distance to near (Scheiman et al. 2005). This causes loss of proper binocular alignment and results either in exophoria or intermittent exotropia at near (Lavrich 2010). The increased accommodative and convergence effort to maintain convergence at near leads to symptoms of asthenopia with near work. Patients can experience blurred vision, horizontal diplopia, the perception of print moving on the page, difficulty maintaining their place on a page while reading, the need to close one eye for reading, epiphora, fatigue, and frontal or periocular headaches. These symptoms of CI may be aggravated by illness, stress, or lack of sleep. There are two peaks in the incidence of convergence insufficiency: (1) high school or college age, when there is an increased demand for near work and (2) early middle age, when the initiation of bifocal use leads to decreased accommodative convergence (Mazow 1971).
The diagnosis of CI is based upon a combination of the symptoms and physical exam findings. A remote near point of convergence (NPC) of greater than 10 cm is a common finding in convergence insufficiency. NPC is a measurement of how close one can bring a fixation target to the nose while maintaining fusion. Normal NPC varies slightly from patient to patient, but is usually less than 10 cm from the nose. Conversely, in CI, the NPC can be increased to 30 cm or more (von Noorden 2002 and Wright et al. 1995). To measure the NPC, the patient fixates on a small target at near. The examiner slowly moves the target closer to the nose until the patient reports diplopia or ocular divergence is noted by the examiner. The distance from the nose to the target is recorded as the NPC.
Patients with CI also have reduced fusional convergence amplitudes, which means that they have a reduced amount of convergence available to overcome temporal disparity in order to maintain binocular fusion at near. Fusional convergence amplitudes are tested at near by having the patient focus on an accommodative target and holding a base out prism bar in front of one eye. Then, the power of the base out prism is gradually increased and the patient is instructed to try to maintain a single image while looking through the prism. The blur point is the power of the prism at which the patient reports that the image becomes blurry. The break point is the power of the base out prism at which diplopia is reported by the patient. Then the prism power is gradually decreased until the patient regains fusion (reports a single image), and this is recorded as the recovery point. Sheard's criterion suggests that for a patient to be comfortable, the fusional amplitudes must be twice the amount of the phoria (Sheard 2005). Normal fusional convergence amplitudes at near have a break point around 36 prism diopters and a recovery point around 32 prism diopters (Mazow 1971).
Another clinical test to help diagnose convergence insufficiency is jump convergence. Jump convergence is a test of the maximum amount a patient can converge comfortably in free space through the prism. Prisms of differing strengths are held in front of the eye and the patient is asked to converge through the prism in order to keep a target single at 33 cm. Jump convergence can be helpful when planning exercises and treatment for CI.
Patients with CI typically demonstrate an exophoria or intermittent exotropia at near, but may also demonstrate orthophoria. This is usually due to accommodative convergence, and these patients may show an increased exophoria at near when +3.00 D spherical lenses are held in front of the eyes to relax convergence at near. With distance fixation, patients may be orthophoric or have a smaller angle exophoria.
CI syndrome must be differentiated from CI-type intermittent exotropia. In CI-type intermittent exotropia, the near deviation is greater than the distance deviation by at least 15 prism diopters. This condition may be associated with true convergence insufficiency syndrome or with a normal NPC and normal fusional convergence amplitudes. Convergence exercises may have an important role in some cases of CI-type intermittent exotropia, but they may be difficult for patients due to temporal suppression. In addition, CI-type intermittent exotropia usually shows a larger deviation at near than CI syndrome does and may require surgical correction.
In addition, it is important to test the near point of accommodation (both monocularly and binocularly), as a combined insufficiency of accommodation and convergence can sometimes occur. In one study, accommodative insufficiency was found in 13 of 42 patients (30.9%) with convergence insufficiency. The rate of comorbidity increases with the severity of the convergence insufficiency (Serna et al. 2011).
CI usually presents with longstanding and chronic complaints of diplopia at near, with a negative past medical history, and a medication list that lacks any medication known to affect accommodation (Scheiman and Wick 2002). The differential diagnosis of CI includes convergence paralysis, which is a comitant supranuclear gaze disorder secondary to ischemic infarction, demyelination, or viral infection. Unlike convergence insufficiency, in convergence paralysis the onset of diplopia or asthenopia is acute. A reduced near point of convergence (meaning a near point of convergence that is greater than 10 cm) has been associated with Parkinson's disease, dorsal midbrain syndrome, and medial rectus weakness from conditions like myasthenia gravis and multiple sclerosis.
Of note, there is an association between CI and attention deficit hyperactivity disorder (ADHD). One study found a higher prevalence of ADHD in patients with CI (9.8%) as compared with the 1.8%-3.3% incidence of ADHD in the general US population (Granet et al. 2005). In addition, they found three times the prevalence of CI in the ADHD population (15.9%) when compared to controls. CI may be a comorbid disorder in patients with ADHD. Conversely, the medications used in the management of ADHD may aggravate CI due to their known side effects of decreased accommodation and increased blurring (Bennett et al. 1999).
Orthoptic convergence exercises are the best treatment for CI. The amount of treatment time necessary is variable between patients. Exercises should be continued until the patient reports an improvement in or resolution of asthenopic symptoms. Patients often need to continue exercises long-term, but less frequently, for maintenance of symptom resolution. Base-in prism glasses can be prescribed to alleviate asthenopic symptoms in presbyopes with CI, or for patients who fail or are unable to complete exercises (Teitelbaum, Pang, Krall 2009).
The first line of treatment for CI is pencil push-up exercises, which are used to reduce or normalize the near point of convergence. To do pencil push-ups, the patient holds an accommodative target at arm's length, focuses on the image to keep it single, and then slowly brings the target toward the nose until diplopia is noted. When diplopia occurs, the patient stops moving the target and refocuses on it until the image becomes clear and single again. The image is then held clearly and singly for 30 seconds. If the patient is unable to make the image single again, the target may be moved away from the nose very slowly until it again becomes a single image, and then the patient may try again. This exercise is done every evening for a few minutes before bed. The goal is to eventually be able to move the target to the nose without diplopia.
These exercises involve having the patient place a small accommodative target on the wall at eye level. The patient will stand at arm's length in front of the target and hold a prism base out in front of one eye. The power of the prism should be the power at which the patient is comfortable fusing in free space (jump convergence). The patient works to keep the target clear and single for 30 seconds and repeats the process a few times before stepping backwards and repeating the exercise. The goal is to continue moving farther from the wall until a single image can be maintained at a distance of ten feet. These exercises should be done in the evening, because they will cause eye fatigue. Prism recession exercises should be continued until the patient reports resolution of asthenopic symptoms.
It is helpful to use a red filter and a light for convergence exercises in patients who are not consistently aware of when their eye diverges, due to suppression. These exercises are done with the patient holding a red filter in front of one eye while looking at a light held at arm's length. The patient should see one light (either pink or red). As the light source is moved closer to the nose, the dissociative red filter will allow the patient to recognize diplopia when one eye diverges. When diplopia occurs, they will either see one light (either the red or the white) disappear or they will see the pink light split into two lights: one red and one white. The patient is made consciously aware of the NPC through this technique. This is the point where they will attempt to hold a single and clear image for 30 seconds. The patient should continue moving the light closer and closer to the nose over time until the light can reach the nose without causing diplopia.
The computer orthoptic program (Computer Orthoptics, Gold Canyon, Arizona, USA) leads the patient through exercises which include saccades, pursuits, base-in and base-out stereograms, and jump convergence exercises. Computer orthoptic exercises offer a game-like interface and are engaging for younger patients.
Three different colored beads are placed equidistant on a 20 cm string. One end of the string is tied to a doorknob and the other end is held against the tip of the patient's nose. The patient focuses first on the distant bead, then the intermediate bead, and lastly the nearest bead, each time making sure to create a clear and single image before changing fixation to the next bead. If the patient has difficulty keeping the near bead as a single image, that bead can be moved farther away from the nose, held for 30 seconds, and then slowly moved closer and closer to the nose until the nose is reached.
A stereogram is made up of two similar but separate images. The patient is given a stereogram card to hold at arm's length, and instructed to hold a pen halfway between the card and his or her face. The patient then converges the eyes in order to focus on the pen in front of the card. Doing this will elicit physiologic diplopia. If the patient is able to appreciate physiologic diplopia, then a third image will appear in the middle of the two pictures on the card. The middle image is a combined image of the two pictures superimposed on top of one another. The goal is to be able to appreciate three clear images on the stereogram card.
EPIDEMIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT
|
Triantafilou DM, Welder JD, Longmuir SQ. Convergence Insufficiency. Dec 10, 2014; Available from: https://eyerounds.org/cases/198-Convergence-insuffic.htm

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.