Coats Disease:
11-year-old male with poor vision in left eye
Coats Disease:
11-year-old male with poor vision in left eye
Chief Complaint: 11-year-old boy who failed a vision screening test at football practice.
History of Present Illness: The patient was asymptomatic until the prior week when at a vision screening for school football he failed to identify any letters on the eye chart using his left eye. After being evaluated by a local vitreoretinal specialist he was referred to the University of Iowa Hospitals and Clinics for further evaluation and treatment.
Past Ocular History: Two years earlier the patient had been told by his school nurse that the vision in his left eye was not as good as the vision in his right eye. He was encouraged to see a local ophthalmologist but did not follow up.
Medical History: No previous medical history.
Medications: None.
Family History: No family history of vision problems.
Social History: Patient resides with his parents and a sibling. He is entering sixth grade.
Ocular Exam:
- Visual Acuity: OD: 20/15; OS: 20/300 PH NI
- Pupils: OD: 5→ 3; OS: 5→4 with >1.8 log unit relative afferent pupillary defect
- Extraocular Motility: Full in diagnostic positions of gaze
- Confrontation Visual Fields: Full OD & OS
- Intraocular Pressure: OD: 21; OS: 17
- External and Anterior Segment Exams: Unremarkable
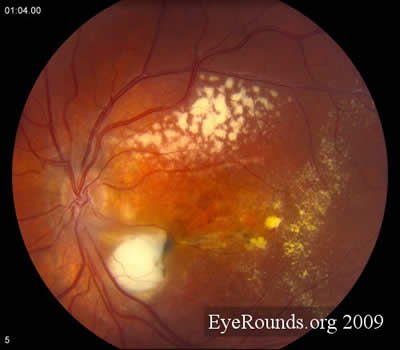
- Dilated Fundus Exam: Unremarkable on the right. On the left there is extensive subretinal fibrosis with areas of exudation throughout the macula and hemorrhage with light bulb aneurysms temporally. There is a fibrous nodule with surrounding subretinal fluid in the inferior macula and additional fluid temporal to the macula.
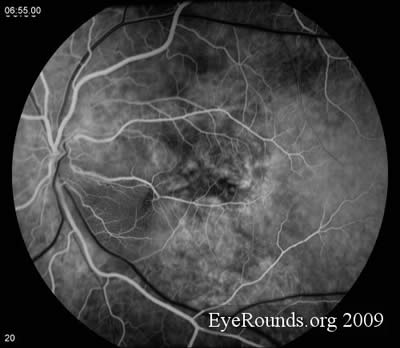
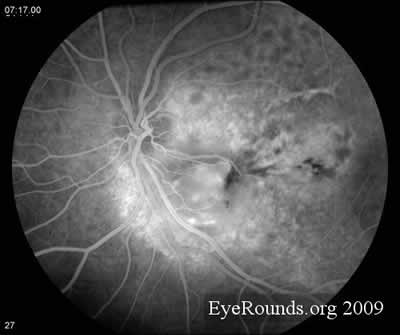
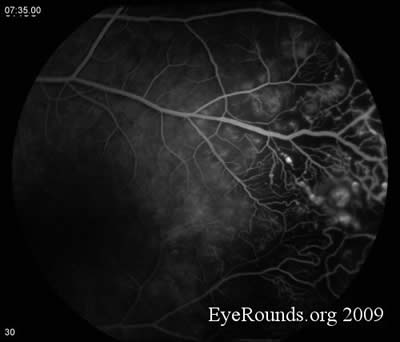
- Fluorescein Angiography: Unremarkable on the right. On the left there is extensive capillary dropout, light bulb aneurysms, and dilated telangiectatic vessels.
- See images.
Treatment: The patient was taken to the operating room for exam under anesthesia and application of 861 spots of laser to the temporal periphery of the left eye. On 3-month follow up, his vision had improved to 20/160 in the left eye; most of the exudate on the left had resolved. There was a small area of saccular aneurysms inferiorly which were treated with laser. At 1-year follow-up the patient’s visual acuity in the left eye had stabilized at 20/200. There were no active aneurysms or hemorrhage.
Discussion: First described in 1908 by the Scottish ophthalmologist George Coats, Coats Disease is a rare eye condition that can cause partial or complete blindness. It is sometimes referred to as exudative retinitis, retinal telangiectasis, or Leber multiple miliary aneurysm disease. Coats Disease is an idiopathic condition characterized by telangiectatic and aneurismal retinal vessels. Increased permeability of these abnormal retinal vessels causes leakage of serum into the intraretinal and subretinal spaces. This fluid can track into the macula causing loss of central vision. Progression is slow, but partial or complete retinal detachment can occur at advanced stages. Visual prognosis depends upon the stage at which the disease is diagnosed.
The progression of Coats Disease can be divided into stages.
- Stage 1 is characterized by abnormal dilation of retinal blood vessels and can be treated with laser photocoagulation or cryotherapy. Vision loss is usually reversible. However, Coats Disease is infrequently diagnosed at Stage 1 because patients are often too young to articulate their symptoms.
- Stage 2 is characterized by both telangiectasia and exudation, and is best treated with laser photocoagulation. Visual prognosis is contingent upon the degree of involvement of the fovea, where foveal sparing portends a favorable prognosis.
- Exudative retinal detachment characterizes Stage 3.
- Total retinal detachment characterizes Stage 4. Visual prognosis at this point is poor, and some eyes diagnosed at this stage will eventually require enucleation.
- Finally, Stage 5 is characterized by irreversible blindness.
Bevacizumab, ranibizumab, and triamcinolone have all been used as adjuncts in the treatment of Coats disease.
There is no evidence to suggest genetic transmission of Coats Disease. In one case, a somatic mutation of the Norrie Disease Protein (NDP) gene located on Xp11.2 was found in an affected individual and in her son, who had Norrie disease. Coats Disease presents unilaterally in 80% of patients and preferentially affects males with a male to female ratio of 3:1. The adolescent form of Coats Disease occurs in patients younger than 16-years-old (average age 5-years-old); an adult form occurs in older patients.
Diagnosis: Coats Disease
EPIDEMIOLOGY
- Also known as exudative retinitis, retinal telangiectasis, or Leber multiple miliary aneurysm disease
- Inheritance pattern unknown
- Very rare; occurs predominately in young males (male:female 3:1)
- Adolescent form (<16 years-old); "Adult" form (>16 years-old)
- 80% unilateral
- Characterized by abnormal retinal blood vessel development
- Can cause poor prognosis in advanced stages
- Retinal detachment at advanced stages
|
SIGNS
- Leukocoria--early warning sign
- "Light-bulb" aneurysms
- Heavy exudates, +/- serous retinal detachment
- Telangiactasia
- Vascular endothelium and pericyte loss
- Vascular mural disorganization; loss of blood-retinal barrier
|
SYMPTOMS
- Blurred vision
- Photopsias and floaters
- Usually painless
|
TREATMENT
- Ablation of causative lesions
- Laser photocoagulation or cryotherapy can be performed
- Steroid and anti-VEGF injections have been successfully used in case series
|
References
- Budning, KS et al. Visual Prognosis of Coats Disease. J AAPOS.1998;2(6):356-359.
- Kanski, Jack J. Clinical Ophthalmology: A Systematic Approach. 6th ed. Elsevier Ltd., 2007.
- Lin, KL et al. Prospects for Treatment of Pediatric Vitreoretinal Diseases with Vascular Growth Fact Inhibition. Seminars in Ophthalmology. 2009;24(2):70-76.
- Shields, JA et al. Classification and Management of Coats Disease: the 2000 Proctor Lecture. Am J Ophthalmology. 2001;31(5):572-583.
- Ridley ME, Shields JA, Brown GC, Tasman W. Coats’ disease: evaluation of management. Ophthalmology. 1982;89:1381-1387.
- Coats G. Forms of retinal disease with massive exudation. Royal London Ophthalmic Hospital Reports.1908;17(3):440-525.
- Davis AE. Coats' Disease of the Retina: Report of Two Cases.Trans Am Ophthalmol Soc. 1921;19:222-9.
- Lin KL, Hirose T, Kroll AJ, Lou PL, Ryan EA. Prospects for treatment of pediatric vitreoretinal diseases with vascular endothelial growth factor inhibition. Semin Ophthalmol. 2009;24(2):70-6.
- Cakir M, Cekiç O, Yilmaz OF. Combined intravitreal bevacizumab and triamcinolone injection in a child with Coats disease. J AAPOS. 2008;12(3):309-11.
- Advanced Coats' disease. Management with repetitive aggressive laser ablation therapy. Schefler AC, Berrocal AM, Murray TG. Retina. 2008;28(3 Suppl):S38-41. Erratum in: Retina. 2009;29(1):127.
- Hsu J, Forbes B, Maguire AM. Total exudative retinal detachment in coats disease: biochemical analysis of the subretinal exudate.Retina. 2006;26(7):831-3.
- Shienbaum G, Tasman WS. Coats disease: a lifetime disease. Retina. 2006;26(4):422-4.
- Couvillion SS, Margolis R, Mavrofjides E, Hess D, Murray TG. Laser treatment of Coats' disease. J Pediatr Ophthalmol Strabismus. 2005;42(6):367-8.
- Smithen LM, Brown GC, Brucker AJ, Yannuzzi LA, Klais CM, Spaide RF. Coats' disease diagnosed in adulthood. Ophthalmology. 2005;112(6):1072-8.
Suggested citation format:
Brinton JP, Jensen LE, Folk JC. Coats Disease: 11-year-old male with poor vision in left eye. EyeRounds.org. Dec. 30, 2009; Available from: http://www.eyerounds.org/cases/100-Coats-Disease.htm.
last updated: 12-30-2009
Image Permissions:

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.